 
There are more ways things can go wrong than right. Importantly, entropy is a state function, like temperature or pressure, as opposed to a path function, like heat or work. the entropy of a random variable is the average level of information, surprise, or uncertainty inherent to the variable’s possible outcomes. Entropy is a measure of how dispersed and random the energy and mass of a system are distributed. The magnitude of the increase is greater than the magnitude of the decrease, so the overall entropy change for the formation of an NaCl solution is positive. Entropy provides a good explanation for why Murphy’s Law seems to pop up so frequently in life. More specifically, we can define it as: Entropy is a scientific concept as well as a measurable physical property that is most commonly associated with a state of disorder. Each hydrated ion, however, forms an ordered arrangement with water molecules, which decreases the entropy of the system. Send us feedback about these examples.\): The Effect of Solution Formation on Entropyĭissolving NaCl in water results in an increase in the entropy of the system. These examples are programmatically compiled from various online sources to illustrate current usage of the word 'entropy.' Any opinions expressed in the examples do not represent those of Merriam-Webster or its editors. Sebastian Smee, Washington Post, After seven years in the Hammerskins, exhaustion and entropy were setting in. Even though thermodynamics itself does not describe processes as a function of time, the second law defines a unique direction of time (times arrow) as the direction in which total entropy increases. Further confusions are produced by some attempts to generalize entropy with similar but not the same concepts in other disciplines. mo.dynamics implies the principle that the total entropy, which is a measure of disorder, must increase steadily. The book’s subtitle is: What we know and what we do not know. 2022 Succumbing to something closer to entropy than evolution, the watercolor starts to puddle and bruise. Entropy is the most used and often abused concept in science, but also in philosophy and society. In 2015, I wrote a book with the same title as this article. Ahmed Almheiri, Scientific American, 17 Aug. 2021 This is the island formula for the entanglement entropy of the Hawking radiation. Conor Feehly, Discover Magazine, 3 Nov. We have a closed system if no energy from an outside source can enter the system. In science, entropy is used to determine the amount of disorder in a closed system. James Riordon, Scientific American, In short, the tendency for systems to move from low entropy to high entropy, the particular spacetime conditions of our solar system and the indeterminacy of the future combine to create our particular conception of time. Entropy is a measure of the amount of energy that is unavailable to do work in a closed system. James Riordon, Scientific American, The expansion allows the universe to smooth out, dissipating the entropy before collapsing again. Quanta Magazine, That flaw is entropy, which builds up as a universe bounces. 2022 If the entropy of the system decreases, the entropy of the environment must increase such that the sum of the two entropies can only increase or stay the same, but never decrease. Jennifer Ouellette, Ars Technica, 2 Dec.  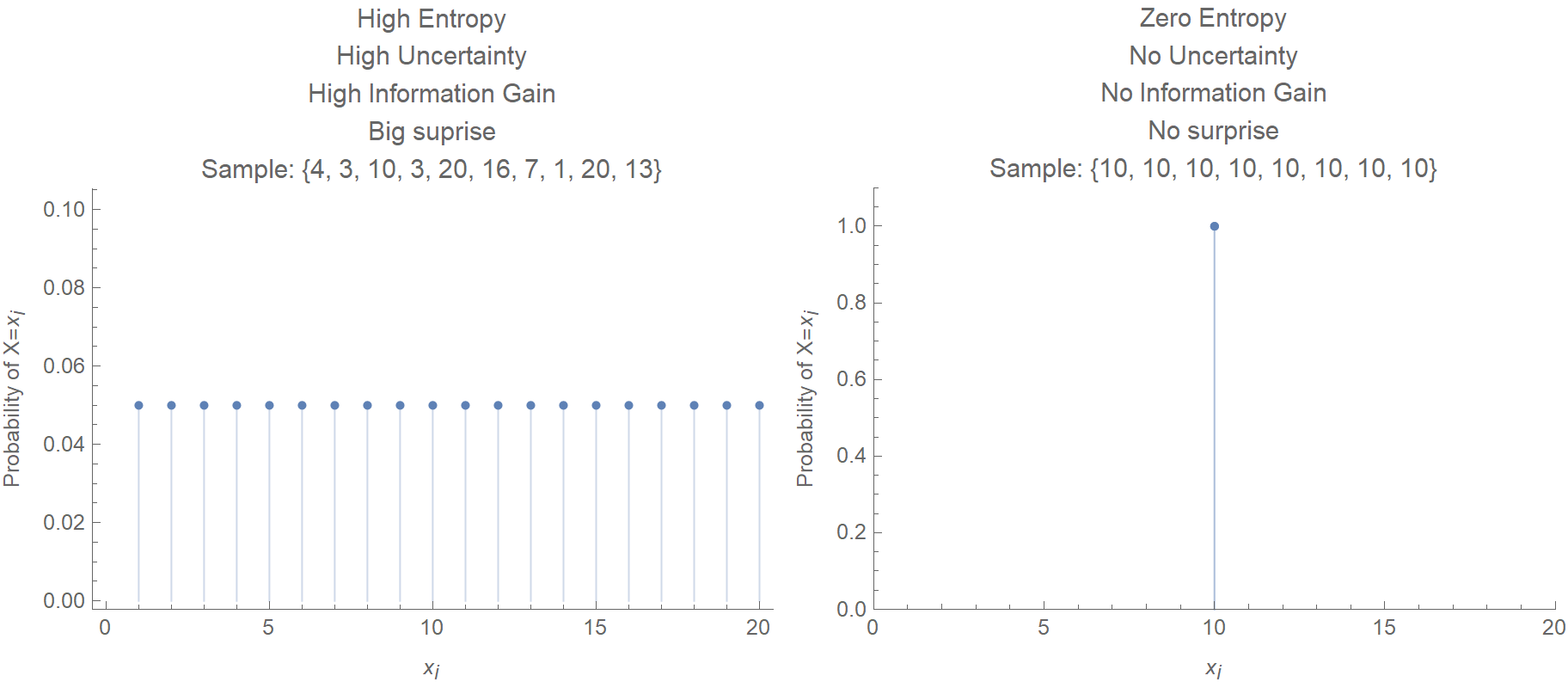
Recent Examples on the Web Jacob Bekenstein realized in 1974 that black holes also have entropy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
